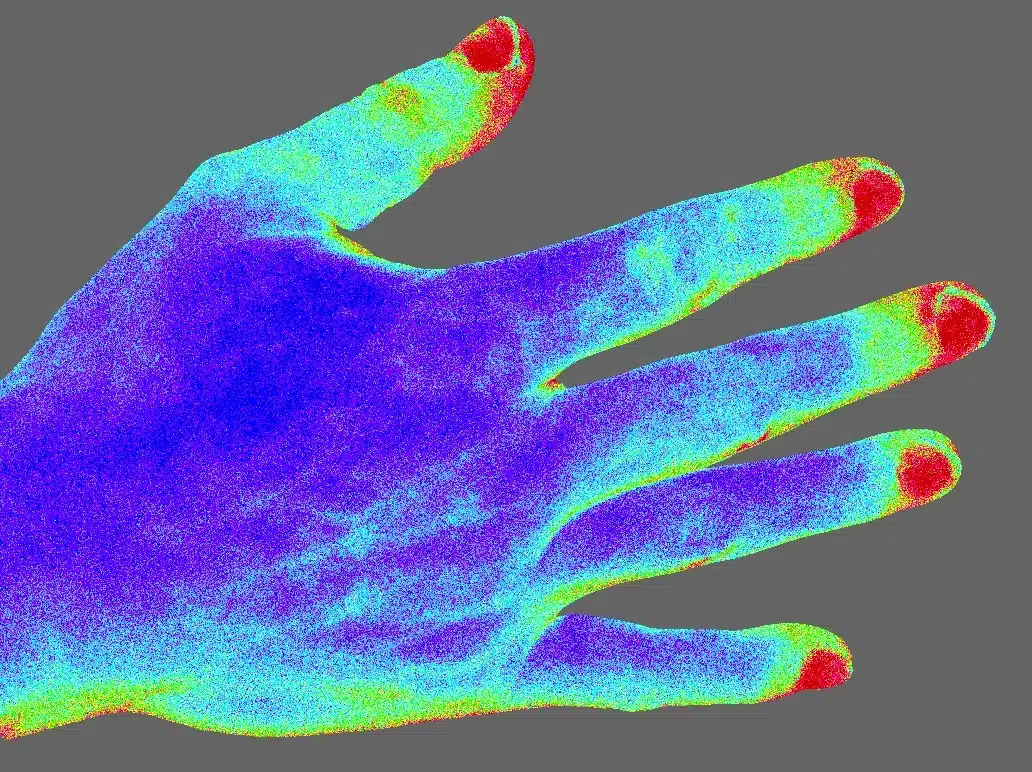
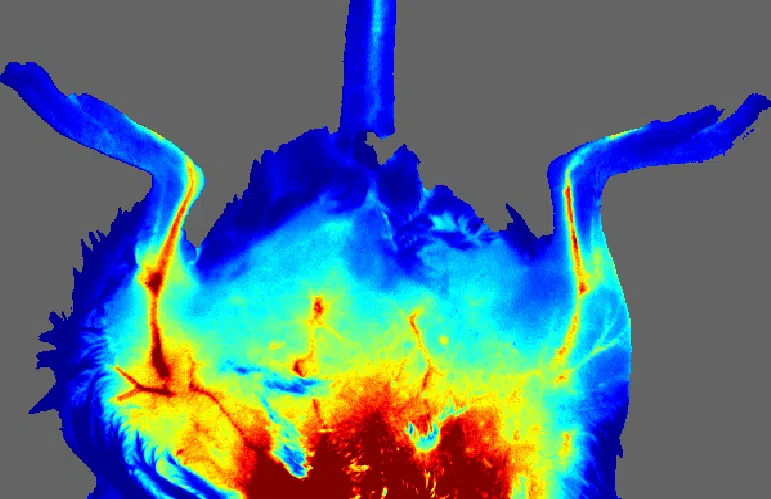
The moorFLPI-2 blood flow imager uses the laser speckle contrast technique to deliver real-time, high-resolution blood flow images, providing outstanding performance in a wide range of pre-clinical and clinical research applications. Optical zoom means you can assess small areas right up to a full size adult hand with a single imager.
Capture every detail with a new unique laser/ single USB camera package. The key specs that matter: pixel resolution of just 3.9 microns (6.6Mpixels/cm2), image sizes ranging from just 6mm x 8mm right up to 225mm x 300mm, using a unique 10x optical zoom function all at up to 100 frames per second…
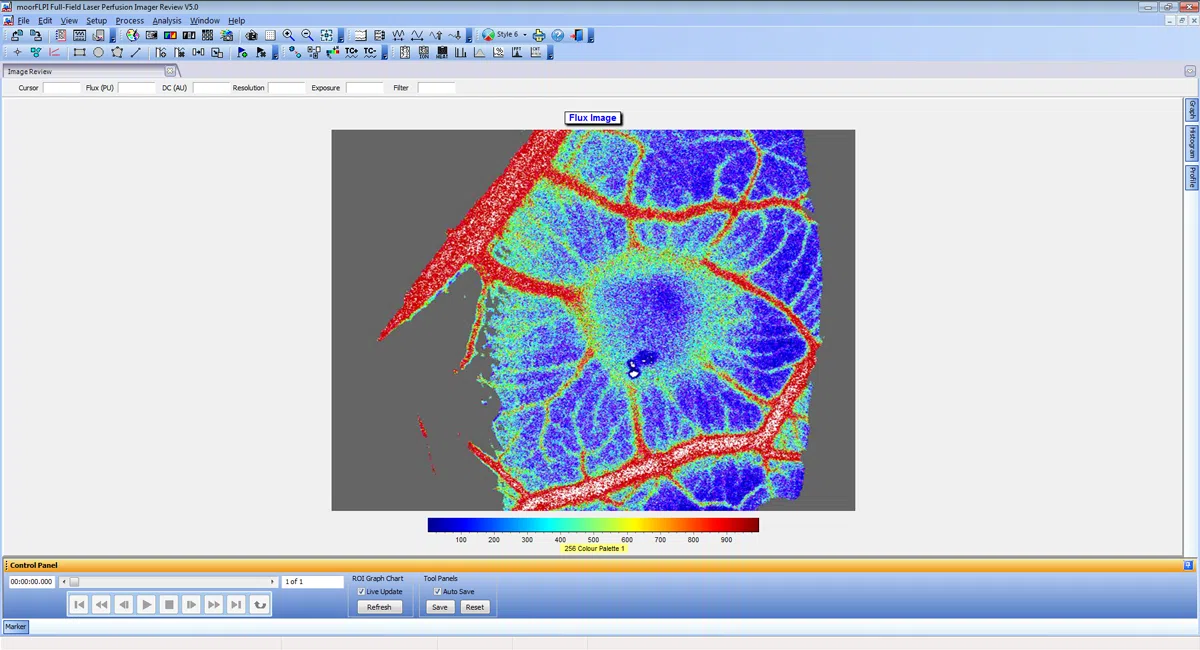
moorFLPI2 features an ergonomically designed scan head and highly refined software package which promotes a smooth workflow and enables the high through-put required to scan cohorts quickly and accurately. Advanced analysis functions help you to draw sound conclusions from your blood flow images. For full details of the software please click here.
moorFLPI-2 highlights include;
- Image any exposed tissue (skin or surgically exposed tissues) and species.
- Easy to use, single USB connection, flexible and simple setup helps get you up and running quickly.
- Three measurement modes with flexible sampling rates (spatial, temporal and sliding window algorithms) to optimise your data for frame rate, spatial resolution and file size.
- Best spatial resolution of 3.9 microns per pixel (6.6Mpixels/cm2).
- Real-time video frames rates – up to 100 frames per second at full field (no windowing).
- Image areas range from 6mm x 8mm to 225mm x 300mm with a unique, motorised 10x optical zoom and auto focus.
- Colour photo and blood flow images provided by a unique single USB3 camera, RGB illumination system. Blood flow and photo images precisely matched.
- Compact design with flexible stand options for laboratory or clinic use.
- Unique protocol control – automation of pressure cuff, tissue heating and iontophoresis protocols with automated reporting, eases workflow and improves accuracy.
- CE marking. Well established in the clinical and research communities for best performance and quality.
- Laser stability hardware for the ultimate in reliable and consistent measurement over minutes, hours and days.
Please contact us, to discuss your specific application requirements with a Moor product specialist. Ask on-line demo to see the new system in action or evaluate it at your own facility.
Please click here for an informative landmark article in the Institute of Physics publication, OLE (Optics and Lasers Europe). The article gives some background to the Full Field technique and offers a comprehensive history, from early beginnings to the current developments at Moor Instruments. Comments are featured from Dr David Briers, the originator of the speckle technique.