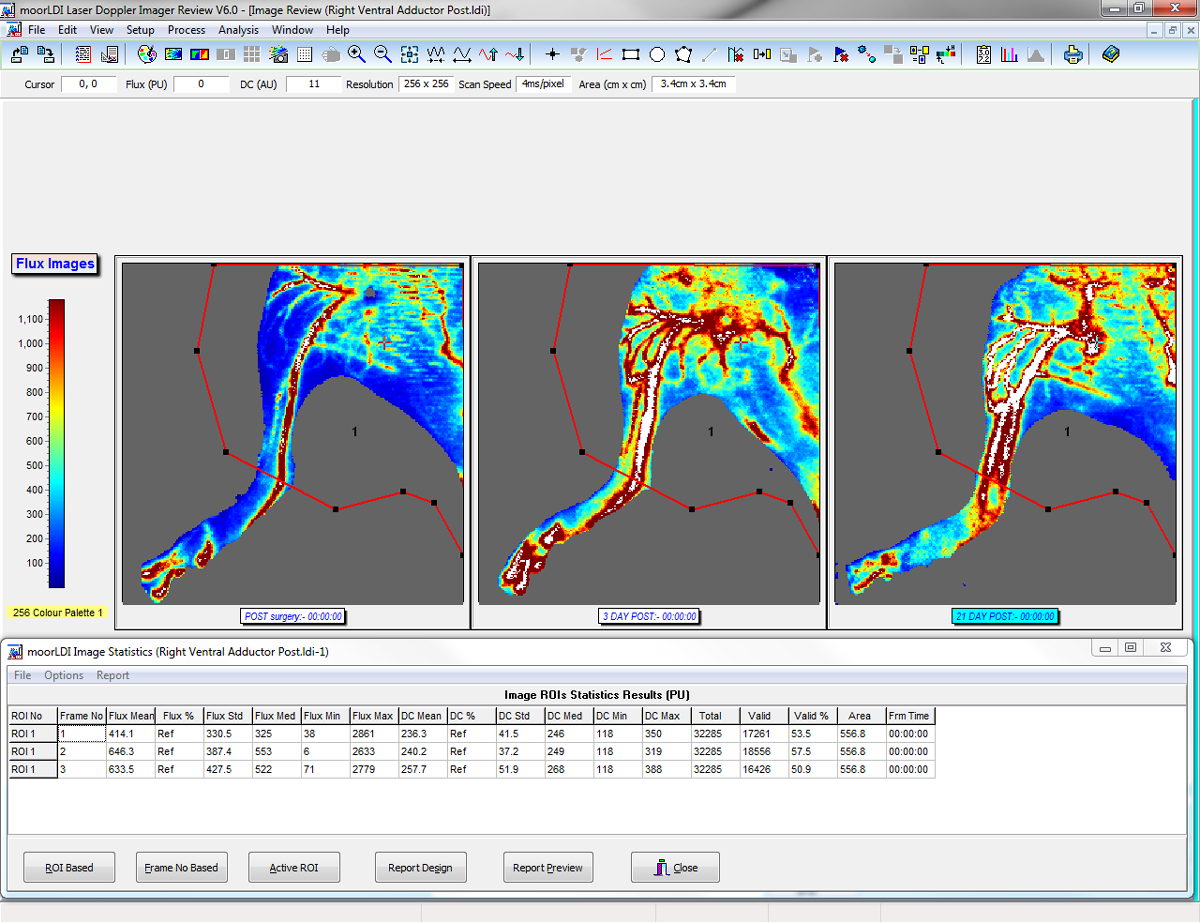
The moorLDI2-HIR high resolution laser Doppler imager is suitable for a wide range of pre-clinical research investigations, more typically where smaller areas are under investigation. The system features unique focused optics to provide 50 micron pixel size and 512 x 512 pixel resolution for high resolution blood flow images. The scan areas range from just 2.5cm x 2.5cm up to 25cm x 25cm with scan times typically less than 5 minutes. Use of a focussed laser provides a deeper measurement depth, optimal for angiogenesis studies such as hind limb ischemia and tumour modelling and pre-clinical cerebral blood flow imaging. Highly refined image measurement and analysis software allows for flexibility in measurement set up and comprehensive analysis functions. The moorLDI2-HIR features a colour photo image of the scanned area and automatic distance measurement, making the positioning and comparison of images easier.
The system is in routine use in numerous laboratories and clinics globally and employs unique, optical design and signal processing in order to generate the highest resolution and clearest images of its class. Laser Doppler imaging (LDI) is often compared to laser speckle imaging and whilst there are some similarities, both techniques offer unique advantages. LDI (and moorLDI2-HIR in particular) generally offers deeper penetration enabling enhanced visualisation of small vessels below the tissue surface, perfect for pre-clinical studies. For certain applications these features are critical.
Other features and benefits include;
- Non contact measurement – painless for patient, aids infection control, no chemical tracers or dyes needed.
- Daylight operation – use in most lab, clinic or theatre settings.
- Flexible scan sizes – from just 2.5cm x 2.5 cm up to 25cm x 25cm.
- High spatial resolution – to catch the finest detail to 50 micron.
- Single and Repeat imaging modes – compare flow from region to region within the same scan and scan the same region repeatedly to assess changes over time.
- Advanced Windows compatible software – to ease setup and scanning. Post Measurement processing functions to make the most of your data.
- Protocol control – set the imager to control flexible tissue heating, pressure cuff control and transdermal drug delivery routines – reproducible, precise and reliable.
- Digital Trigger In/ Out – to synchronise with external devices.
- Digital Signal Processing and high quality optics – providing the highest sensitivity to changes in blood flow and superb reliability.
- Choice of stands – for benchtop use.
NOTE: If you are interested in clinical research and larger scan areas please consider the moorLDI2-IR large area imager or the moorFLPI-2 laser speckle imager.